This section provides guidance on monitoring persons with chronic hepatitis C virus (HCV) infection who are starting direct-acting antiviral (DAA) treatment, are on treatment, or have completed therapy. The section is divided into 4 parts:
- Pretreatment and on-treatment assessment and monitoring (including persons with incomplete adherence)
- Posttreatment follow-up for persons in whom treatment failed to clear HCV
- Posttreatment follow-up for those who achieve a sustained virologic response (SVR; virologic cure)
- Additional considerations if treatment includes ribavirin.
Pretreatment and On-Treatment Monitoring
| Recommended Assessments Prior to Starting Direct-Acting Antiviral Therapy | |
| RECOMMENDED | RATING |
| Staging of hepatic fibrosis is essential prior to HCV treatment (see Testing and Linkage to Care and When and in Whom to Treat). FIB-4 index is sufficient to identify persons with and without advanced fibrosis/cirrhosis for the purpose of selecting a DAA regimen. | I, C |
| Assessment of potential drug-drug interactions with an individual’s concomitant prescription medications, over-the-counter medications, and herbal supplements is required prior to starting DAA therapy. When possible, an interacting co-medication should be stopped or switched to an alternative with less risk for potential interaction during HCV treatment. (See Table of Drug Interactions with Direct-Acting Antivirals and Selected Concomitant Medications below or use an online resource such as University of Liverpool interaction checker.) | I, C |
| Health care practitioners should educate persons who will receive HCV therapy about DAA medication administration (eg, dose, frequency, food coadministration requirements, management for missed doses, potential adverse events), the crucial importance of adherence, and the need to inform their health care practitioners about any changes to their medication regimen. | I, C |
The following laboratory tests are recommended within 6 months prior to starting DAA therapy:
|
I, C |
| The safety of ribavirin-free DAA regimens in humans has not been established during pregnancy or for nursing mothers. Counseling should be offered to women of childbearing age before beginning HCV treatment. (See pregnancy recommendations below.) | I, C |
All persons initiating DAA therapy should be assessed for active hepatitis B virus (HBV) coinfection with HBV surface antigen (HBsAg) testing, and for evidence of prior infection with HBV core antibody (anti-HBc) and HBV surface antibody (anti-HBs) testing.
|
IIa, B |
| All persons should be assessed for HIV coinfection prior to initiating DAA therapy. | IIa, B |
| Testing for the presence of resistance-associated substitutions (RASs) prior to starting treatment should be performed as recommended in the Initial Treatment and the Retreatment sections. Additional information about RAS testing can be found in the HCV Resistance Primer. | IIb, B |
Persons scheduled to receive an HCV NS3 protease inhibitor (ie, grazoprevir, voxilaprevir, glecaprevir) should be assessed for a history of decompensated liver disease and liver disease severity using the Child-Turcotte-Pugh (CTP) score.
|
I, A |
| a Unlike the AASLD/IDSA HCV guidance, the AASLD guidelines for treatment of chronic hepatitis B utilizes the GRADE system to rate recommendations. Please see that document for further information about its rating system. | |
Recommended Monitoring During HCV Antiviral Therapy |
|
|---|---|
| RECOMMENDED | RATING
|
| Clinic visits or telephone contact are recommended as clinically indicated during treatment to ensure medication adherence and monitor for adverse events and potential drug-drug interactions, especially with newly prescribed medications. See table of Drug Interactions with Direct-Acting Antivirals and Selected Concomitant Medications. | I, B |
| Inform persons taking diabetes medication of the potential for symptomatic hypoglycemia. On-treatment and posttreatment monitoring for hypoglycemia is recommended. | I, C |
| Inform persons taking warfarin of the potential for changes in their anticoagulation status. On-treatment and posttreatment INR monitoring for subtherapeutic anticoagulation is recommended. | I, C |
| Persons receiving elbasvir/grazoprevir should be monitored with a hepatic function panel at 8 weeks and again at 12 weeks if receiving 16 weeks of treatment. | I, B |
| A ≥10-fold increase in ALT values from baseline at any time during treatment should prompt discontinuation of DAA therapy (especially with signs or symptoms of liver inflammation or increasing conjugated bilirubin, alkaline phosphatase, or INR). An increase in ALT <10-fold from baseline that is accompanied by any weakness, nausea, vomiting, jaundice, or significantly increased bilirubin, alkaline phosphatase, or INR should also prompt discontinuation of DAA therapy. Asymptomatic increases in ALT <10-fold from baseline should be closely monitored with repeat testing at 2-week intervals. If levels remain persistently elevated, consideration should be given to discontinuation of DAA therapy. |
I, B |
| Quantitative HCV RNA testing (viral load) is recommended 12 or more weeks after completion of therapy to document sustained virologic response (SVR), which is consistent with cure after HCV infection. |
I, B |
| Evaluation of SVR at four weeks (SVR4) by quantitative HCV RNA testing (viral load) can be considered an alternative measure of HCV cure among people without cirrhosis or prior DAA exposure. |
II, A |
For individuals who are positive for HBsAg and not already receiving HBV suppressive therapy because their baseline HBV DNA level does not meet treatment criteria, one of two approaches may be taken:
|
IIa, B |
The recommended pretreatment testing assumes that a decision to treat with DAA medications has already been made and that the testing involved in deciding to treat—including testing for HCV genotype (if unable to use pangenotypic DAAs) and FIB-4 or other assessment of hepatic fibrosis—has already been completed (see When and in Whom to Initiate HCV Therapy).
Fibrosis staging is an important component of pretreatment assessment; many noninvasive modalities are available for clinical use (Sterling, 2025); (Sterling, 2024). Yet delaying HCV treatment for additional fibrosis assessment beyond FIB-4 may present additional barriers for patients and result in compromising retention in care. A retrospective cohort study conducted in New Orleans noted a median time of 6.4 months to complete direct hepatic fibrosis assessment (defined as liver ultrasound, transient elastography, proprietary blood-based testing, or biopsy) following HCV diagnosis in 791 people (Jones, 2023). A study of 5 federally qualified health centers in Philadelphia demonstrated that 20.4% of persons evaluated by an HCV-treating clinician did not subsequently complete hepatic fibrosis assessment by liver ultrasound, transient elastography, or proprietary blood-based testing (Coyle, 2019). As noted in the simplified HCV treatment pathway, FIB-4 assessment is sufficient for the selection and initiation of HCV treatment in treatment-naive individuals as it provides acceptable diagnostic performance for detecting advanced fibrosis/cirrhosis among people with chronic hepatitis C infection (Patel, 2025); (Sterling,2025). FIB-4 scores >3.25 indicate advanced hepatic fibrosis/cirrhosis; surveillance for hepatocellular carcinoma and decompensation should be performed. A calculator to determine FIB-4 can be found here.
Health care practitioners must assess each individual’s medications for potential drug-drug interactions before prescribing DAAs. Resources include the manufacturer’s prescribing information and other drug-drug interaction websites (eg, the University of Liverpool). The table below lists known drug-drug interactions between HCV DAAs and selected medications.
Drug Interactions with Direct-Acting Antivirals and Selected Concomitant Medications
| Concomitant Medications | SOF/VEL | GLE/PIB | SOF/VEL/VOX | LDV/SOF | EBR/GZR |
|---|---|---|---|---|---|
|
Acid-reducing agents |
Antacids |
H2RA |
Antacids |
Antacids |
|
|
Alpha-1 blockers |
Prazosin |
Prazosin |
Prazosin |
Silodosin |
Prazosin |
|
Analgesics |
Metamizole |
Alfentanil |
Metamizole |
|
Fentanyl |
|
Antiarrhythmics |
Amiodarone |
Amiodarone |
Amiodarone |
Amiodarone |
Amiodarone |
|
Digoxin |
Digoxin |
Digoxin |
|||
|
Anticoagulant and antiplatelet agents |
Apixaban |
Dabigatran |
Dabigatran |
Apixaban |
Apixaban |
|
Apixaban |
Apixaban |
||||
|
Anticonvulsants and barbiturates |
Amobarbital |
Amobarbital |
Amobarbital |
Amobarbital |
Amobarbital |
|
Rufinamide |
Rufinamide |
Rufinamide |
Rufinamide Zonisamide |
Rufinamide |
|
|
Antihypertensives |
Diltiazem |
Aliskiren |
Aliskiren |
Aliskiren |
Eplerenone |
|
Enalapril |
|||||
|
Antimycobacterials |
Rifabutin |
Rifabutin |
Rifabutin |
Bedaquiline |
Rifabutin |
|
Bedaquiline |
Rifabutin |
Bedaquiline |
|||
|
Antipsychotics – first generation |
|
Pimozide |
|
Pimozide |
Pimozide |
|
Droperidol |
|||||
|
Antipsychotics – second generation |
|
Aripiprazole |
Paliperidone |
Paliperidone |
Aripiprazole |
|
Antiretrovirals |
See HIV/HCV Coinfection Section |
||||
|
Azole antifungals |
|
Ketoconazole |
|
|
Ketoconazole |
|
Benzodiazepines |
|
|
|
|
Midazolam |
|
Bronchodilators |
|
Theophylline |
|
|
|
|
Buprenorphine/ naloxone |
|
|
|
|
|
|
Calcineurin inhibitors |
|
Cyclosporine |
Cyclosporine |
|
Cyclosporine |
|
Tacrolimus |
Tacrolimus |
||||
|
Cancer Therapies |
Acalabrutinib |
Acalabrutinib |
Acalabrutinib |
Acalabrutinib |
Acalabrutinib |
|
Vinblastine |
Imatinib |
||||
|
|
|
|
|
||
|
Cholesterol-lowering agents |
Atorvastatin |
Atorvastatin |
Atorvastatin |
Rosuvastatin |
Atorvastatin |
|
Ezetimibe |
Ezetimibe |
Atorvastatin |
|||
|
Cisapride |
|
|
|
|
|
|
COVID-19 antivirals |
Molnupiravir |
Molnupiravir |
Molnupiravir |
Molnupiravir |
Molnupiravir |
|
Nirmatrelvir/ |
Nirmatrelvir/ |
Nirmatrelvir/ |
|||
|
Ergot derivatives |
|
|
|
|
|
|
Ethinyl estradiol containing products |
|
|
|
|
|
|
Glucocorticoids |
|
Dexamethasone |
Dexamethasone |
|
Dexamethasone |
|
Heart failure agents |
Bosentan |
Bosentan |
Bosentan |
|
Bosentan |
|
Ambrisentan |
Ambrisentan |
Ambrisentan |
|||
|
Herbals |
St. John’s wort |
St. John’s wort |
St. John’s wort |
St. John’s wort |
St. John’s wort |
|
Loop diuretics |
|
|
|
|
|
|
Macrolide antimicrobials |
Troleandomycin |
Erythromycin |
Erythromycin |
Telithromycin |
Telithromycin |
|
Troleandomycin |
Troleandomycin |
Troleandomycin |
|||
|
Phosphodiesterase-5 inhibitors |
|
|
|
|
|
|
Recreational Drugs |
|
Carfentanil |
|
|
Carfentanil |
|
H2RA, histamine H2 antagonist; PPI, proton pump inhibitor; DHP CCB, dihydropyridine calcium channel blocker; non-DHP CCB, non-dihydropyridine calcium channel blocker. Green indicates coadministration is safe; yellow indicates a dose change or additional monitoring is warranted; and red indicates the combination should be avoided. Specific concomitant medications or medication classes with actual or theoretical potential for interaction are listed in the box. |
|||||
The education of persons initiating HCV treatment and their caregivers about potential adverse effects of DAA therapy and their management is an integral component of treatment and is important for a successful outcome in all patient populations. During DAA treatment, individuals should be followed at clinically appropriate intervals to ensure medication adherence, assess adverse events and potential drug-drug interactions, and monitor blood test results necessary for patient safety (Drazilova, 2018); (Abdel Alem, 2017); (Rindone, 2017); (Pavone, 2016); (DeCarolis, 2016); (Soriano, 2016).
Routine testing for HCV RNA during treatment is not recommended unless the ALT level fails to decline (when elevated) or there are concerns regarding adherence with DAA treatment. There are no data to support stopping treatment based on detectable HCV RNA during the first 4 weeks of treatment or that detectable HCV RNA at this time point signifies medication nonadherence.
During clinical trials with elbasvir/grazoprevir, with or without ribavirin, 1% of participants experienced ALT elevations from normal levels to >5 times the upper limit of normal, generally at or after treatment week 8. ALT elevations were typically asymptomatic, and most resolved with ongoing therapy or completion of therapy. Higher rates of late ALT elevations occurred in females, those of Asian descent, and persons aged ≥65 years. Hepatic laboratory testing should be performed prior to therapy, at treatment week 8, and as clinically indicated. For persons receiving 16 weeks of therapy, additional hepatic laboratory testing should be performed at treatment week 12 (Zepatier prescribing information, 2021).
A cohort study evaluated the acute liver injury related to protease inhibitor-based DAA therapy versus non-protease inhibitor-based therapy. A total of 18,498 initiators of protease inhibitor-based DAA therapy (paritaprevir/ritonavir/ombitasvir ± dasabuvir, elbasvir/grazoprevir, or glecaprevir/pibrentasvir) matched 1:1 on propensity score to initiators of non-protease inhibitor-based therapy (ledipasvir/sofosbuvir or sofosbuvir/velpatasvir). The study was conducted within the US Veterans Health Administration from 2014 through mid-2019. The investigator tracked acute liver injury outcomes during exposure to DAA therapy—specifically, the incident development of: 1) ALT >200 U/L; 2) severe hepatic dysfunction, defined as coagulopathy (INR ≥1.5) with hyperbilirubinemia; and 3) hepatic decompensation according to baseline FIB-4 score (≤3.25 versus >3.25). The analysis demonstrated that the risk of incident ALT elevations was increased among protease inhibitor-based DAA initiators in both FIB-4 groups. However, the risk of severe hepatic dysfunction or hepatic decompensation did not differ between participants who received protease inhibitor-based DAA therapy compared with those treated with non-protease inhibitor-based therapy in either FIB-4 group. (Torgersen, 2021).
People being treated with amiodarone should not receive sofosbuvir-based regimens due to risk of life-threatening arrhythmias. Because of its long half-life, it is advised that persons should be off amiodarone for at least 6 months before initiating sofosbuvir. If the decision is made to start sofosbuvir in this setting, continued vigilance for bradycardia should be exercised.
Simplified HCV Treatment for Treatment-Naive Adults
Data from a global sample of persons undergoing antiviral treatment for chronic HCV infection suggested that a minimal monitoring approach was safe and achieved SVR at a rate comparable to that with standard monitoring. This minimal monitoring approach was examined in a phase 4, open label, single-arm trial that enrolled 400 treatment-naive participants aged ≥18 year with HCV RNA >1,000 IU/mL from Brazil, South Africa, Thailand, Uganda, and the US with capped inclusion of compensated cirrhosis and HIV/HCV coinfection (Solomon, 2022). Pregnancy, breastfeeding, or evidence of chronic HBV infection (HBsAg positive) were exclusion criteria. However, participants with resolved HBV infection—hepatitis B core total antibody (anti-HBc) with or without positive hepatitis B surface antibodies (anti-HBs)—were eligible. Participants initiated treatment with fixed dose sofosbuvir (400 mg)/velpatasvir (100 mg) once daily for 12 weeks. Minimal monitoring involved: 1) no pretreatment genotyping; 2) dispensing the entire treatment course (84 tablets) at entry; 3) no scheduled visits or laboratory monitoring; and 4) remote contact at week 4 to assess DAA adherence and at week 22 to schedule SVR assessment at week 24.
Of the 400 participants, 399 initiated sofosbuvir/velpatasvir treatment. At entry, 166 (42%) were living with HIV, 34 (9%) had compensated cirrhosis, and 121 (32%) of 374 with an available HBV panel (HBsAg, anti-HBc, anti-HBs) had evidence of resolved HBV infection. Overall, 379 of the 399 who initiated treatment achieved SVR (95.0%; 95% CI 92.4–96.7%). A total of 14 (4%) of 397 participants reported serious adverse events between treatment initiation and week 28 but none were treatment-related or led to treatment discontinuation or death (Solomon, 2022).
Given the findings of this minimal monitoring study, simplified HCV treatment approaches are available for HCV treatment-naive adults without cirrhosis d for HCV treatment-naive adults with compensated cirrhosis. Because of the possible risk for HBV reactivation that could be fulminant, persons with HBV/HCV coinfection should be excluded from simplified HCV treatment approaches.
Assessment of HCV Cure
It is essential to test for HCV RNA after DAA treatment completion. Undetectable or nonquantifiable HCV RNA 12 weeks or longer after treatment completion defines sustained virologic response (SVR12), which is consistent with cure of chronic HCV infection. Virologic relapse is rare 12 weeks or longer post treatment completion (Sarrazin, 2017); (Simmons, 2016).
A substantial number of people can be lost to care after completing DAA therapy and prior to the week 12 posttreatment completion benchmark thereby compromising practitioners’ ability to evaluate for HCV cure. A national study of 41,102 commercially insured persons treated for HCV between 2014 and 2019 revealed that only 62.7% completed an SVR12 assessment (Ferrante, 2022). Similarly, a study including 906 Oklahoma Medicaid members treated for chronic HCV infection between 2014 and 2016 found that only 40.6% underwent SVR12 assessment (Pham, 2018).
Data from randomized, controlled trials of first-line DAAs have demonstrated a high correlation between SVR4 and SVR12, such that >99% of people who achieve SVR4 will subsequently achieve SVR12 (Gane, 2021); (Sulkowski, 2021); (Bernstein, 2014). This correlation has been demonstrated predominantly among people without cirrhosis or prior DAA exposure. Achievement of SVR4 can therefore be considered an alternative measure of HCV cure and should be considered among persons without cirrhosis and without prior DAA exposure for whom there may be potential barriers to completion of an SVR12 assessment. Repeat quantitative HCV RNA testing can be considered beyond SVR4 for persons who experience an ALT increase above the upper limit of normal.
Pregnancy and Nursing Mothers
Few adequate and well-controlled human studies are available to establish whether DAAs pose a risk to pregnancy outcomes or whether DAAs and their metabolites are present in breastmilk. An open-label, phase 1 study of pregnant persons without HIV infection and with chronic genotype 1 infection evaluated a 12-week course of ledipasvir/sofosbuvir initiated between 23 weeks to 24 weeks of gestation (Chappell, 2020). Among 7 evaluable participants, all achieved SVR12. Adverse events related to ledipasvir/sofosbuvir were less than or equal to grade 2. All 7 participants delivered at term with undetectable HCV viral loads at delivery. In the 12-month follow-up period, the growth parameters and trajectories, and the neurodevelopmental assessments were normal for all the infants (Chappell, 2020).
Given the dearth of data on this topic, clinicians should discuss with persons who have childbearing capacity that DAAs should be used during pregnancy only if the potential benefit of DAA therapy justifies the potential risk of harm to the fetus. The health benefits of DAA therapy for nursing mothers should be weighed against the health benefits of breastfeeding and the possible adverse effects of the DAA regimen on the breastfed child. Given the relatively short duration of treatment and the availability of ribavirin-free regimens for most patients, the potential risk of harms and benefits of delaying pregnancy until HCV DAA therapy is completed should be considered. See the HCV in Pregnancy section for additional information.
Reactivation of Hepatitis B Virus Infection
Cases of HBV reactivation, occasionally fulminant, during or after DAA therapy have been reported in persons with HBV/HCV coinfection who were not receiving HBV suppressive therapy (Mücke, 2018); (Bersoff-Matcha, 2017); (Chen, 2017). Therefore, all persons initiating DAA therapy should be assessed for HBV coinfection with HBsAg testing and for evidence of prior infection with anti-HBc and anti-HBs testing. HBV vaccination is recommended for all susceptible individuals. Testing for HBV DNA should be performed prior to DAA therapy in persons who are HBsAg positive. HBsAg positivity does not represent a contraindication to DAA therapy. Persons meeting criteria for treatment of active HBV infection should be started on HBV therapy simultaneously or before initiation of DAA therapy (Terrault, 2018).
Persons with a low or undetectable HBV DNA level can either receive prophylactic HBV treatment for the duration of DAA treatment until assessment for SVR12 or be monitored at regular intervals (usually not more frequently than every 4 weeks) for HBV reactivation with HBV DNA testing. If monitoring is selected, HBV treatment should be started if the HBV DNA level increases >10-fold or is >1000 IU/mL in a person with undetectable or unquantifiable HBV DNA prior to DAA treatment. There are insufficient data to provide clear recommendations for the monitoring of HBV DNA among persons who test positive either for anti-HBc alone (isolated anti-HBc) or for both anti-HBc and anti-HBs (resolved infection). However, the possibility of HBV reactivation should be considered in these individuals in the event of an unexplained increase in liver aminotransferase levels during and/or after completion of DAA therapy.
Posttreatment Follow-Up for Persons in Whom Treatment Failed
Recommended Monitoring for Persons in Whom Treatment Failed to Achieve a Sustained Virologic Response |
|
|---|---|
| RECOMMENDED | RATING
|
| Retreatment for chronic HCV is recommended utilizing the regimens recommended in the Retreatment section | I, C |
| Disease progression assessment every 6 months to 12 months with a hepatic function panel, complete blood count, and international normalized ratio is recommended for persons who are either not retreated or for whom a second or third DAA treatment course fails. | I, C |
| Surveillance for hepatocellular carcinoma with liver ultrasound examination (with or without alpha fetoprotein) every 6 months is recommended for persons with cirrhosisa in accordance with the AASLD guidance on the diagnosis, staging, and management of hepatocellular carcinoma | Low, Conditionalb |
| For persons with cirrhosis, endoscopic surveillance for varices should be performed in accordance with the AASLD guidance on portal hypertension bleeding in cirrhosis | Guidanceb |
| a Fordecompensated cirrhosis, please refer to the appropriate section. b Unlike the AASLD/IDSA HCV guidance, the AASLD guidelines use the GRADE system to rate recommendations. Please see that document for further information about this rating system. |
|
The Following Monitoring Is Not Recommended During or After Therapy |
|
|---|---|
| NOT RECOMMENDED | RATING
|
| Monitoring for HCV drug resistance-associated substitutions (RASs) during or after therapy is not recommended unless retreatment will be performed. RAS testing is recommended in advance of retreatment therapy. See the Retreatment section for recommendations regarding RAS testing prior to retreatment. Additional information about RAS testing can be found in the HCV Resistance Primer | IIb, C |
People who do not achieve SVR retain the possibility of continued liver injury, progression of hepatic fibrosis, and the potential to transmit HCV infection to others. Such persons should be considered for retreatment per the Retreatment of Persons in Whom Prior Therapy Has Failed section.
Given that persons in whom treatment failed remain at risk for ongoing liver injury and liver fibrosis progression (Dienstag, 2011), these individuals should be monitored for signs and symptoms of decompensated cirrhosis. Persons in whom antiviral therapy failed may harbor viruses that are resistant to 1 or more of the antivirals at the time of virologic breakthrough (Lawitz, 2014a); (Schneider, 2014). There is no evidence to date, however, that the presence of resistance-associated substitutions (RASs) results in more progressive liver injury than would have occurred if the person did not have resistant viruses. Additional information about RASs and RAS testing can be found in the HCV Resistance Primer section. If there remains uncertainty regarding the applicability of RAS testing, consultation with an expert regarding the treatment of HCV infection may be useful.
Posttreatment Follow-Up for Persons Who Achieved a Sustained Virologic Response
Recommended Follow-Up for Persons Who Achieved a Sustained Virologic Response (SVR) |
|
|---|---|
| RECOMMENDED | RATING
|
| For persons without cirrhosis, recommended follow-up is the same as if they were never infected with HCV. | I, B |
| Assessment for HCV recurrence is recommended only if the person develops unexplained hepatic dysfunction, or annual assessment if the person has ongoing risk factors for HCV infection. In such cases, a quantitative HCV-RNA test (viral load) rather than an HCV antibody test is recommended to assess for HCV recurrence. | I, A |
| Surveillance for hepatocellular carcinoma is recommended for persons with cirrhosis,a in accordance with the AASLD guidance on the diagnosis, staging, and management of hepatocellular carcinoma | Strong, Moderateb |
| For persons with cirrhosis, upper endoscopic surveillance is recommended in accordance with the AASLD guidance on portal hypertension bleeding in cirrhosis | Guidanceb |
| Assessment for other causes of liver disease is recommended for persons who develop persistently abnormal liver tests after achieving SVR | I, C |
| a Fordecompensated cirrhosis, please refer to the appropriate section. b Unlike the AASLD/IDSA HCV guidance, the AASLD guidelines use the GRADE system to rate recommendations. Please see that document for further information about this rating system. |
|
Persons with undetectable serum HCV RNA as assessed by a sensitive polymerase chain reaction (PCR) assay ≥12 weeks after treatment completion are deemed to have achieved SVR (ie, cure). The likelihood of achieving SVR with DAA therapy among adherent, immunologically competent, persons who are treatment naive with compensated liver disease generally exceeds 95%. Among persons who achieved SVR with peginterferon/ribavirin treatment, more than 99% have remained free of HCV infection when followed for 5 years after treatment completion (Manns, 2013). Thus, achieving SVR is considered a virologic cure of HCV infection. SVR typically aborts progression of liver injury with regression of liver fibrosis in most (but not all) treated persons (Morgan, 2013); (Morisco, 2013); (Morgan, 2010); (Singal, 2010); (George, 2009). Liver fibrosis and liver function test results improve in most persons who achieve SVR (Morgan, 2013); (Morisco, 2013); (Morgan, 2010); (Singal, 2010); (George, 2009). Because of lack of progression, persons without cirrhosis who achieve SVR should receive standard medical care that is recommended for individuals who were never infected with HCV unless they remain at risk for non-HCV–related liver disease, such as steatotic liver disease or alcoholic liver disease.
Among persons with cirrhosis who achieve SVR, decompensated liver disease (with the exception of HCC) rarely develops during follow-up and overall survival is prolonged (Morgan, 2013); (Morisco, 2013); (Morgan, 2010); (Singal, 2010); (George, 2009). Bleeding from esophageal varices is rare after SVR (Morgan, 2013); (Morisco, 2013); (Morgan, 2010); (Singal, 2010); (George, 2009). Persons with cirrhosis should undergo surveillance endoscopy every 2 years if known to have small varices and every 3 years in the absence of known varices in accordance with AASLD guidance on portal hypertension bleeding (Garcia-Tsao, 2017).
Importantly, persons with cirrhosis remain at risk for developing HCC and should, therefore, undergo surveillance for HCC every 6 months utilizing ultrasound (with or without AFP testing) despite the lowered risk that results from viral eradication (Marrero, 2018). Although multiple studies of persons with cirrhosis who achieved SVR with peginterferon/ribavirin reported a reduction in the risk of developing HCC (Morgan, 2013); (Morisco, 2013); (Morgan, 2010); (Singal, 2010); (George, 2009) and a meta-analysis of persons achieving SVR with DAAs found that the HCC risk did not exceed that seen in persons who experienced SVR with interferon-based treatment after adjustment for baseline risk factors for HCC (Waziry, 2017b). One report found a higher than expected frequency of HCC in persons with HCV-related cirrhosis despite successful DAA treatment (Reig, 2016). However, a prospective observational study of 3,045 persons with cirrhosis found an adjusted hazard ratio for HCC of 0.57 (95% CI 0.40–0.81) following DAA-based therapy, implying a 43% reduction in HCC incidence (Carrat, 2019).
Bleeding from esophageal varices is uncommon after SVR (Morgan, 2013); (Morisco, 2013); (Morgan, 2010); (Singal, 2010); (George, 2009). Nevertheless, persons with compensated cirrhosis who achieve SVR should continue to receive endoscopic surveillance for esophageal varices, in accordance with the AASLD guidance on portal hypertension bleeding (Garcia-Tsao, 2017). Current AASLD recommendations for persons with compensated cirrhosis without known varices is surveillance endoscopy every 2 years if there is evidence of ongoing liver injury from associated conditions, such as obesity or alcohol use, and every 3 years if liver injury is quiescent, such as after alcohol abstinence. Individuals with compensated cirrhosis and known varices should undergo surveillance endoscopy annually if there is evidence of ongoing liver injury from associated conditions, such as obesity or alcohol use, and every 2 years if liver injury is quiescent, such as after alcohol abstinence.
People in whom SVR is achieved but who have another potential cause of liver disease (eg, excessive alcohol use, metabolic syndrome with or without steatotic liver disease, or iron overload) remain at risk for hepatic fibrosis progression. It is recommended that such individuals be educated about the risk of liver disease and monitored for liver disease progression with periodic physical examination, blood tests, and potentially, tests for liver fibrosis by a liver disease specialist.
Persons who achieve SVR can have HCV recurrence due to reinfection or late relapse (Sarrazin, 2017); (Simmons, 2016). A systematic review suggests 5-year recurrence risks of 1%, 11%, and 15% in low-risk HCV monoinfected, high-risk HCV monoinfected (ie, people who currently or formerly injected drugs, incident imprisonment, or men who have sex with men), and persons with HIV/HCV coinfection, respectively (Simmons, 2016). At least annual testing for HCV reinfection among persons with ongoing risk for HCV infection (eg, injection drug use or high-risk sexual exposure) is recommended. A flare in liver aminotransferase levels should prompt immediate evaluation for HCV reinfection (see Management of Acute HCV Infection). Clinicians must test for HCV recurrence using an assay that detects HCV RNA (ie, quantitative or qualitative HCV RNA test) because HCV antibody remains positive in most persons after achieving SVR.
Acute liver injury is common among individuals receiving chemotherapy or immunosuppressive agents. Testing for hepatitis viruses should be included in the laboratory assessment of the cause of acute liver injury in these individuals. Approximately 23% of persons with active HCV infection—especially those with a hematologic malignancy—experience a flare in their HCV RNA level (>10-fold) during chemotherapy. An ALT level increase is less common and clinical symptoms of hepatitis are uncommon (Torres, 2018). Among persons who have recovered from HCV infection, either spontaneously or with DAA treatment, reactivation of HCV infection (ie, detectable HCV RNA) during chemotherapy is distinctly uncommon and is not anticipated to occur since there is no residual reservoir for the virus. Thus, in this latter group, routine testing for HCV RNA during immunosuppressive treatment or prophylactic administration of DAAs during immunosuppressive treatment is not recommended.
Additional Considerations If Treatment Includes Ribavirin
Ribavirin dosage must be adjusted for reduced renal function. Ribavirin causes hemolysis and its long half-life perpetuates the anemia that may occur. People receiving ribavirin should have hemoglobin levels checked during treatment, often after 2 weeks. Clinicians may elect to reduce the ribavirin dose reduced if the person’s hemoglobin decreases by 2g/dL in a 2-week to 4-week time frame or if the individual develops anemia, often defined as hemoglobin <10 g/dL.
Ribavirin causes fetal death and fetal abnormalities in animals. Ribavirin should not be administered to pregnant persons or to those who might become pregnant during or for 6 months after completing ribavirin treatment. Similarly, ribavirin may cause birth defects in offspring of women whose partner was receiving ribavirin when conception occurred. In the rare instances when ribavirin is used, it is imperative for persons of childbearing potential to use at least 2 reliable forms of effective contraception during treatment and for a period of 6 months thereafter. It is recommended that the health care practitioner document the discussion and the individual’s understanding of the potential teratogenic effects of ribavirin in their medical record.
Incomplete Adherence
There are limited data regarding the outcome of persons who have incomplete adherence to DAA therapy or the threshold level of adherence below which the incidence of SVR12 is significantly reduced. Many individuals report missing doses of DAAs. A secondary analysis of data from electronic blister packs that recorded the date and time of each dose among 103 participants in a velpatasvir/sofosbuvir clinical trial demonstrated that approximately one-third (32%) of study participants had <90% adherence (ie, nonadherence) (Cunningham, 2018). The most common episodes of nonadherence lasted 1 to 2 days (61% of episodes); 11% of episodes lasted ≥7 days. Despite the nonadherence, SVR12 was 94% among both DAA adherent (≥90% of doses) and nonadherent (<90% of doses) participants. Longer durations of missed treatment, however, may affect SVR. A study of persons receiving DAA treatment found that only 50% (2/4) of participants with F0-F3 disease who took <4 weeks of their course of therapy experienced SVR, compared with an SVR rate of 99% (109/110) for those who received ≥4 weeks of therapy. Among participants with cirrhosis, a lower SVR12 rate was observed in those who took <8 weeks of therapy compared with participants who took ≥8 weeks (83% [25/30] versus 95% [209/221]) (Fabbiani, 2021). A multicenter US randomized, controlled trial of 496 people who inject drugs evaluated SVR rates by adherence to 12 weeks of sofosbuvir/velpatasvir, measured via electronic blister packs. The median adherence was 75%, and 92.7% achieved SVR12. Participants with ≥50% days of sofosbuvir/velpatasvir adherence achieved an SVR12 rate of >90% (Heo, 2024).
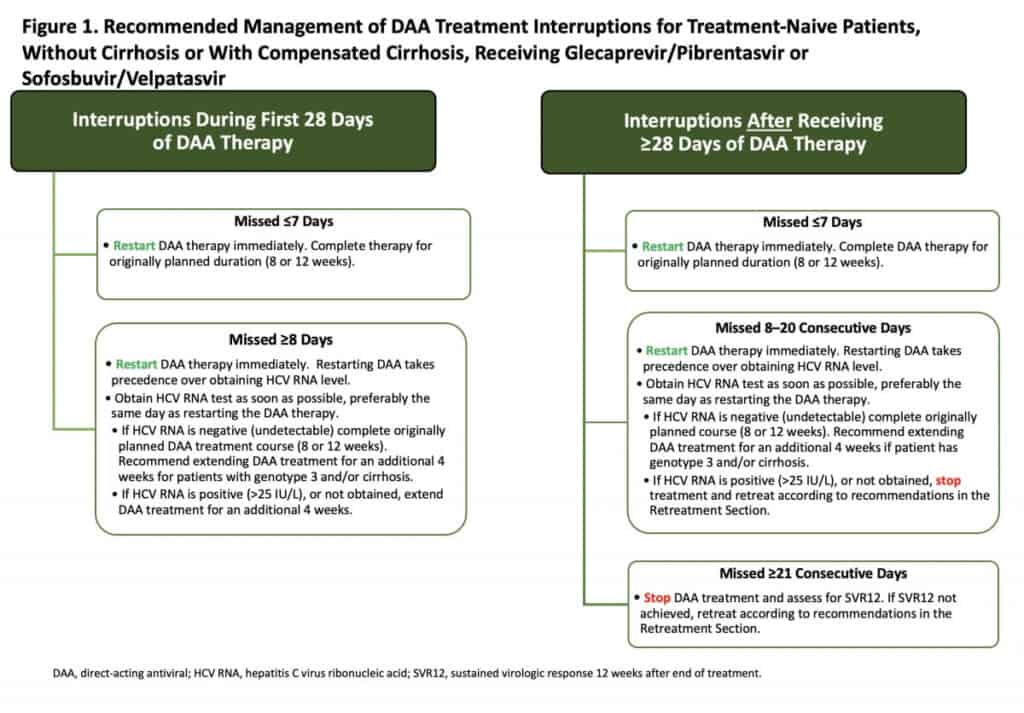
Few data exist on which to base recommendations regarding how to manage persons who have discontinued DAAs for several days to weeks. The recommendations shown in Figure 1 are applicable to treatment-naive persons with acute or chronic HCV, without cirrhosis or with compensated cirrhosis, receiving either glecaprevir/pibrentasvir or sofosbuvir/velpatasvir. These recommendations are based on the opinion of the AASLD/IDSA HCV treatment guidance panel.
Individuals with prior DAA treatment, or receiving other DAA treatment regimens, or other populations (eg, persons who are post transplant or have decompensated cirrhosis) should be managed in consultation with an expert. All persons with incomplete adherence should be asked about factors contributing to adherence or nonadherence and counseled regarding the importance of adherence. In general, the panel considers a treatment interruption of <7 days unlikely to impact SVR12 based on adherence and outcome data from the SIMPLIFY study (Cunningham, 2018).
Click the graphic to enlarge Figure 1.